
 Share This Page
Share This Page| Home | | Sailing | | Alaska 2010 | |  |  |  Share This Page Share This Page |

Copyright © 2010, P. Lutus. All rights reserved. Message Page
| Prior years: |
Alaska 2002 |
Alaska 2003 |
Alaska 2004 |
Alaska 2005 Alaska 2006 | Alaska 2007 | Alaska 2008 | Alaska 2009 |
(double-click any word to see its definition)
Anyone who doubts the truth of the Second Law of Thermodyanamics should try owning a boat. A short description of the Second Law would be: "... an expression of the universal principle of decay observable in nature." Another way to describe it is "entropy."


During my around-the-world sail 25 years ago, I watched every electronic and mechanical system fail, some over and over again. An the end of that four-year voyage, I realized there was nothing on board my boat more complicated than a mousetrap that hadn't been replaced at least once.
I bought my present boat in 2001, and since then about half of everything has failed — engine failures, electronic failures, mechnical failures, plumbing failures. Plumbing failures? — yes, you heard me right — if you look at a pipe on a boat, it's reasonable and prudent to ask, "what flows through that pipe, and will it cause problems when the pipe inevitably bursts and sprays its contents all over the place?"
Some boat equipment failures are inevitable, arising from limitations of materials, however well-chosen. Others result from unebelievably stupid choices made by boat builders. This page describes a stupid, and very common, choice made by boat builders.
Figure 1 shows a boat water heater as it might appear before being installed, already one of the worst-designed pieces of equipment ever to make its way onto a boat, and one that dramatizes the importance of scientific and technical education.
Figure 2 shows a typical boat water heater installed and in servce. Notice that the fittings are brass. Now look up at Figure 1 again — see the one brass fitting? That's a signal from the factory that brass is all right (except it isn't). What can we conclude from this? It means each and every conection to a typical water heater attaches one kind of metal to another.
This brings us the the issue of galvanic corrosion: "... an electrochemical process in which one metal corrodes preferentially when in electrical contact with a different type of metal and both metals are immersed in an electrolyte."Galvanic corrosion is a very well-understood process in which atoms of one metal, in contact with atoms of another and with "an electrolyte" (ordinary water will do), will disintegrate. The solution is to avoid the use of dissimilar metals in boat plumbing — very simple. Can we get boat builders to do this? No, of course not, that would be too easy (Figure 2 — all brass fittings).
Even though I knew what galvanic corrosion was, I didn't think my boat's builders would be so stupid as to ignore it, and I didn't look very closely at my boat's water heater, at least for the first few years. But one night, while at anchor somewhere in Alaska, I heard a mechanical clunk from below, and the fresh water pump began to run. And run, and run.
I turned off the pump and began to investigate. I quickly discovered that one of the brass fittings had simply fallen off the heater, just as though there was nothing holding it on. And at that point, there wasn't — on examining the inside of the fitting, I saw almost no metal remaining from the pipe to which the brass fitting has been attached. It was as though the pipe had dissolved.
But, preoccupied with taking bear pictures and hanging out in pretty Alaska, I decided that someone at the factory had just not attached the heater's fitting properly, and this would be a one-time failure. I purchased a pipe die at a local hardware store, rethreaded the heater pipe, and reattached the brass fitting. Mistake number one.
A year later, another fitting fell off in the same way. I already had made up my mind what this meant, so I just rethreaded that pipe and reattached the fitting as before. Mistake number two.
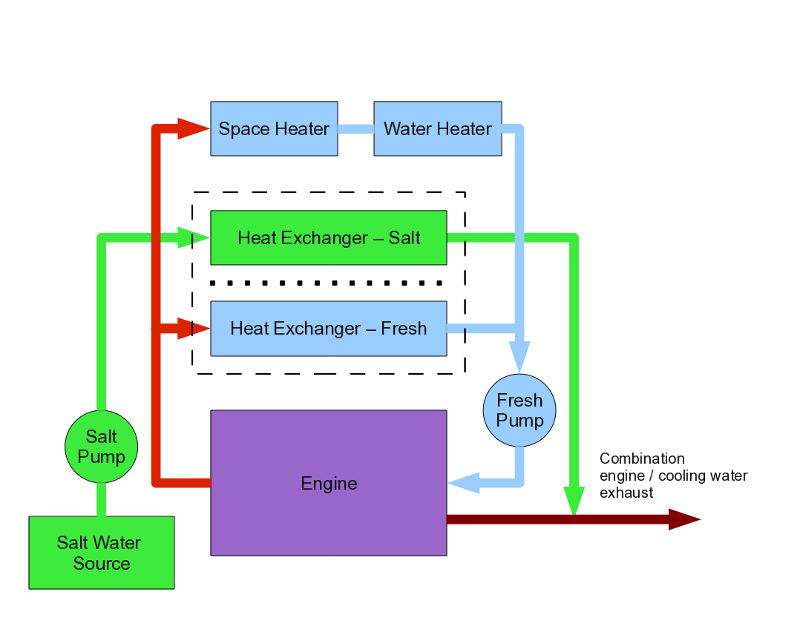
But this season, something much worse happened, something that forced me to rethink my assumptions. Look at Figure 3 on this page — this is the engine's cooling system diagram, with all the extra gadgets that are attached to it for one reason or another. A boat water heater normally has a loop to receive engine cooling water, to heat water while underway (this is why there are so many plumbing fittings on the tank in in Figure 1).But notice something — if the loop that includes the water heater in Figure 3 should fail, the engine's cooling water will spill out into the bilge, the engine will overheat, and you must shut down the engine no matter where you are.
This season, as I left the town of Hoonah one nice morning, the engine overheated and I shut it down. I was just off Cannery Point in a brisk wind that would eventually put me on the beach, so I started problem-solving more quickly than usual. So an overheat alarm, check cooling water — none! Where did the water go? I used a teakettle to repeatedly refill the cooling reservoir so I could get back into the marina, then started tracing out the plumbing system (which led to the first rough sketch of Figure 3).
Unfortunately, the water heater was the last thing I considered, because all its fitting looked sound. But several hours later I conducted an experiment — I started the engine, then crawled around the bilge looking for leaks. Almost immediately I saw a miniature fountain spurting out from one of the brass fittings that routed the engine cooling water through the heater.
Right away I thought how annoying that someone would not be able to attach two pipes together more securely than that. I put on some gloves (185° water!) and grabbed the brass fitting, thinking I would give it a quarter-turn and everything would be all right again. But instead, the fitting fell off the tank and the cooling water ran out into the bilge.
I grabbed the other fittings, pulled, and most of the fittings fell off the tank with the smallest effort. I then saw that I had been explaining away the heater problems as the sloppy work of some anonymous boat builder, but it was now clear that the problem all along had been ... galvanic corrosion. I then said something to myself that I have more occasions to say as I get older: "you are such an idiot."
For those boat owners who wonder how much time they have left before they have the same problem, I can only say if you see brass fittings on the outside of your boat water heater, and if you are at all concerned about boat safety, chances are you are already out of time.
As I write I am awaiting the arrival of a new heater, and I have a plan — I am going to use all plastic fittings and eliminate the brass. All the boat's plumbing is plastic, including the engine cooling water loop, and the only things that weren't plastic were the six brass fittings threaded onto the heater itself — really a brilliant design choice.
An obvious objection to this approach is that typical plastics will melt at a temperature not far above the boiling point of water — won't the plastic fittings melt and cause a different kind of failure if the heater overheats for some reason? Well, yes, that's possible, but I had a chance to run a test after the failure described above — I replumbed the engine cooling loop by attaching the engine cooling water hoses to a couple of ordinary plastic fittings, and threaded those fittings onto the tank. Remember this water is 185° f. I didn't have any problems for the remainder of the season.Also, if the plastic fittings fail, I can replace them for a few dollars. When brass fitting fail, you replace the tank, not the fittings.
I want to add one technical note — if water doesn't flow through a particular brass fitting, no corrosion seems to take place. Look at Figure 1 again — see the brass fitting that is normally already in place from the factory? That's a safety valve, meant to keep the tank from exploding if the thermostat should fail and allow the water to boil. My brass safety valve was one of the exceptions to the pattern of corrosion, and it has to do with the fact that water isn't flowing through it (I think flowing water efficiently carries away the ionized metal atoms, which greatly speeds the corrosion rate).
Here is my advice to avoid this problem in new boat designs and repairs — avoid dissimilar metals in plumbing. That might sound like obvious advice, but almost no one heeds it. Use plastic or something else nonmetallic, or use plumbing fittings all of the same kind of metal. Obviously if suppliers of boat heaters wanted to solve this problem, they would offer high-temperature plastic fittings, or carbon fiber, or ceramic — any of dozens of suitable materials that will not corrode.
But if they did that, they wouldn't be able to sell you a new water heater every few years.
| Home | | Sailing | | Alaska 2010 | |  |  |  Share This Page Share This Page |